by Katrina Rogers | Nov 14, 2019 | Uncategorized
What’s in Module 2 of the CTD? (Note: This post is part of a multi-part series on the CTD this fall) Module 2 of the CTD contains summaries of information presented in detail in modules 3-5. It starts with a one-page introduction that covers the pharmacologic class,...

by Katrina Rogers | Nov 12, 2019 | Uncategorized
IMDRF Draft Guidance is Available for Comment until December 2, 2019 The International Medical Device Regulators Forum released a draft guidance on cybersecurity in early October, and it will be available for public comment until December 2nd. The guidance reinforces...
by Katrina Rogers | Nov 7, 2019 | Uncategorized
Module 1: Regional Administrative Information and Prescribing Information Because the eCTD is based on the ICH CTD, my next few posts will cover the content in each of the specific CTD modules. Module 1 contains the region-specific information and is the only module...
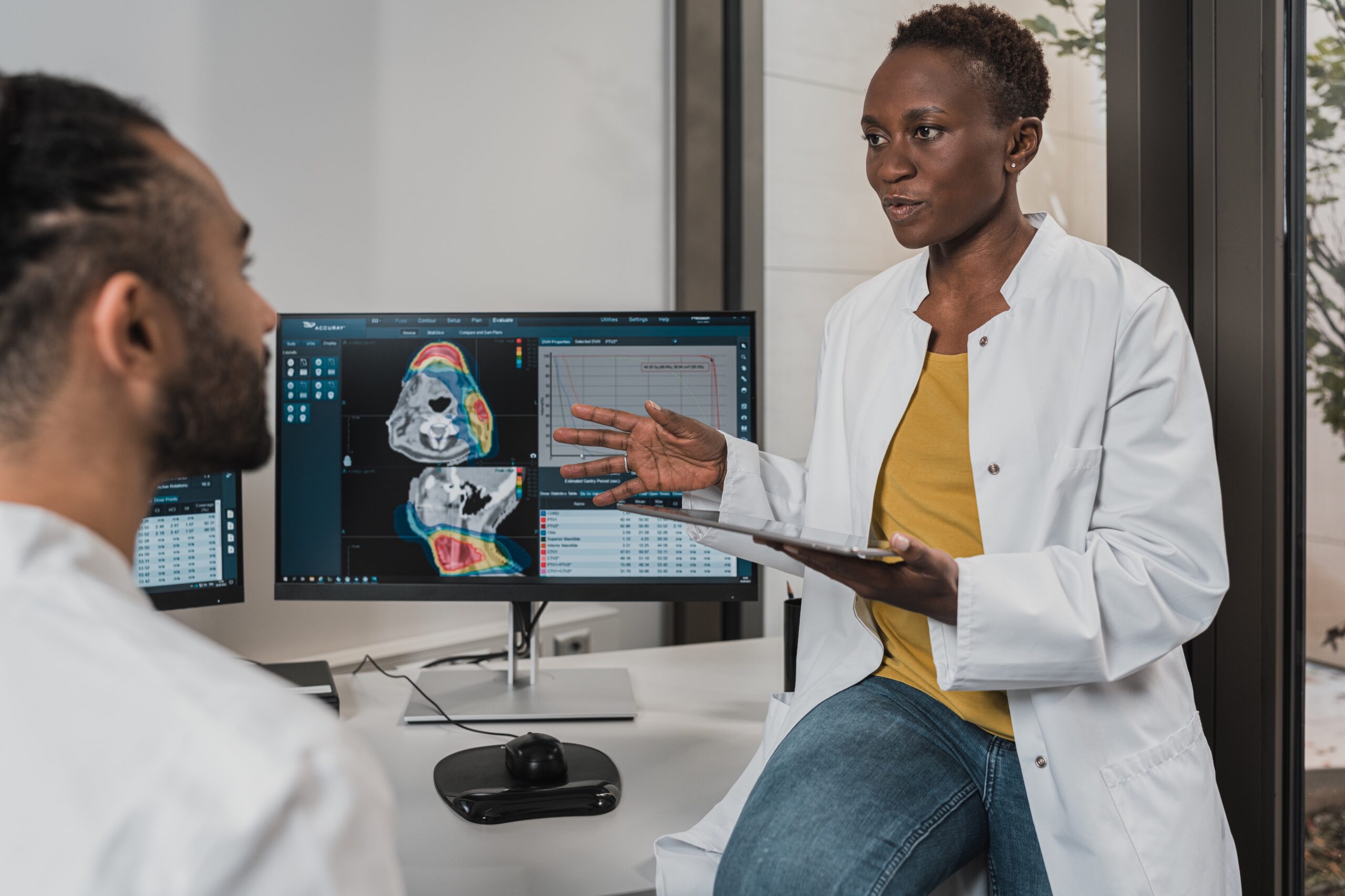
by Katrina Rogers | Nov 5, 2019 | Medical Products, Medicine
CDRH posted seven new modules on the CDRH Learn web page at the end of September/beginning of October. These new modules are: Under The Basics How is CDRH Structured? Is My Product a Medical Device? Under How to Study and Market Your Device Introduction to the...
by Katrina Rogers | Oct 31, 2019 | Uncategorized
The History of the CTD and eCTD (Note: This post is part of a multi-part series on the CTD this fall) The history of the ICH eCTD is shown in this ICH slide deck from 2011, a program overview presented by the Rapporteur (that’s the person appointed to report the...



Recent Comments