by Katrina Rogers | Mar 24, 2020 | COVID
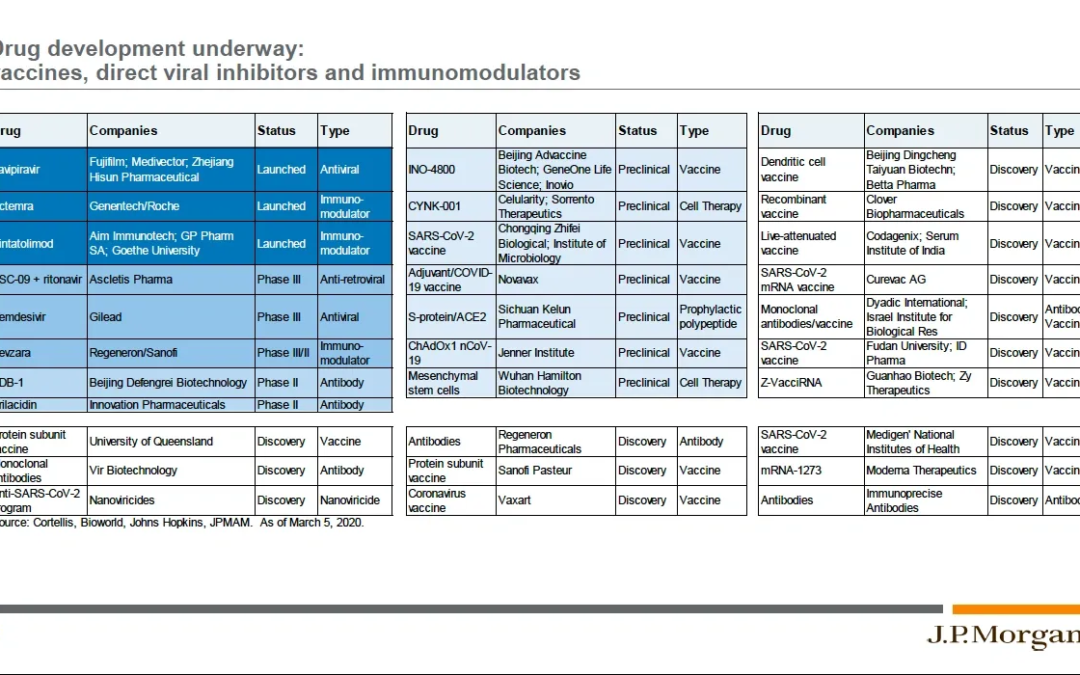
Chapter 6 – Biomedical Mobilization Against COVID-19 The response of our biomedical community to the novel coronavirus outbreak has been impressive, and is illustrated in this slide from a JP Morgan webcast on COVID-19 and the Markets held on March 20th...

by Katrina Rogers | Mar 19, 2020 | Medical Products, Medicine, Product Development
Regulatory bodies worldwide make use of consensus standards developed by independent organizations to establish best practices and conceptual harmony on specialized areas of expertise. Standards organizations for drugs include the regional Pharmacopeia USP, EP, and JP...

by Katrina Rogers | Mar 17, 2020 | Medical Products
Traditionally, pharmacopoeia is a book of medicinal drugs with their effects and directions for use. Today, the pharmacopoeia are regional organizations devoted to creating and maintaining standards to ensure public health. Part of that mission includes maintaining a...

by Katrina Rogers | Mar 12, 2020 | Product Development
If you are involved in the development or manufacturing of a medical product, sooner or later, you will face a regulatory inspection. The individual regulatory agencies publish many of their inspection procedures online, and these documents are valuable references for...
by Katrina Rogers | Mar 10, 2020 | Product Development
With the recent approval of three new next generation sequencing diagnostic tests for tumor profiling, CDRH also announced their approach to regulating such tests. There are three levels of validated biomarkers addressed in each of the market authorizations for the...







Recent Comments