by Katrina Rogers | Aug 26, 2018 | Uncategorized
Companies who are using eCTD for submissions or developing eCTD tools should take note that the ICH M8 group completed an update to the ICH eCTD v4.0 Implementation Package during the ICH June 2018 meeting. The updated implementation package (v1.3) should be posted on...
by Katrina Rogers | Aug 23, 2018 | Uncategorized
Good data standards enable the effective sharing and comparison of scientific data. For example, the international drug regulatory agencies use a consensus standard called eCTD to help them receive, process, review, and archive submission data more effectively. Data...
by Katrina Rogers | Aug 21, 2018 | Uncategorized
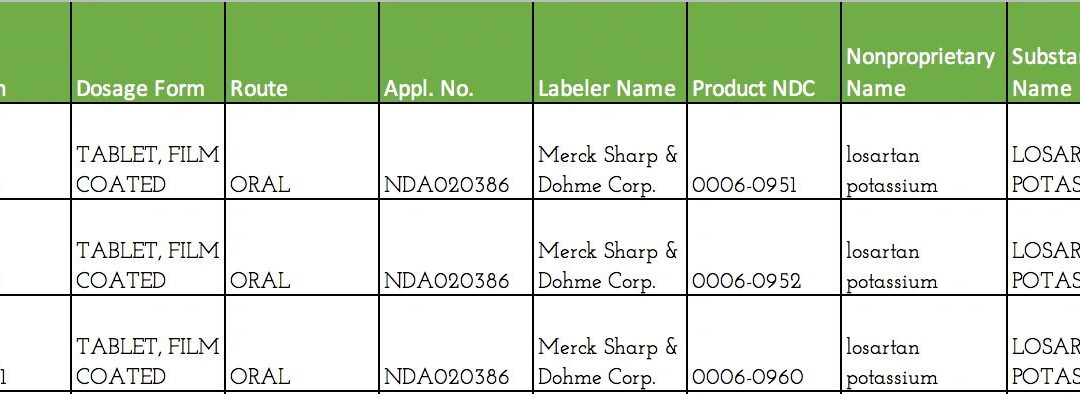
Example NDC codes The FDA has announced a public hearing on 4 approaches to gather comments on proposed changes to the current National Drug Code (NDC). The NDC is a 10- or 11-digit universal product identifier for human drugs in the United States, quickly identifying...
by Katrina Rogers | Aug 19, 2018 | Uncategorized
Last week’s FDA approval of Natural Cycles, a mobile medical app, makes available in the US a personal and discreet form of birth control that’s been available to EU women since August 2017. The app uses early morning temperature measurements logged by the user to...
by Katrina Rogers | Aug 16, 2018 | Uncategorized
The FDA approved Onpattro last week, the first approved medicine to use small interfering RNA (siRNA) to prevent formation of disease causing proteins in the body. This class of therapeutics, known more generally as RNAi, is expected to create breakthroughs in the...



Recent Comments