by Katrina Rogers | Oct 1, 2018 | Uncategorized
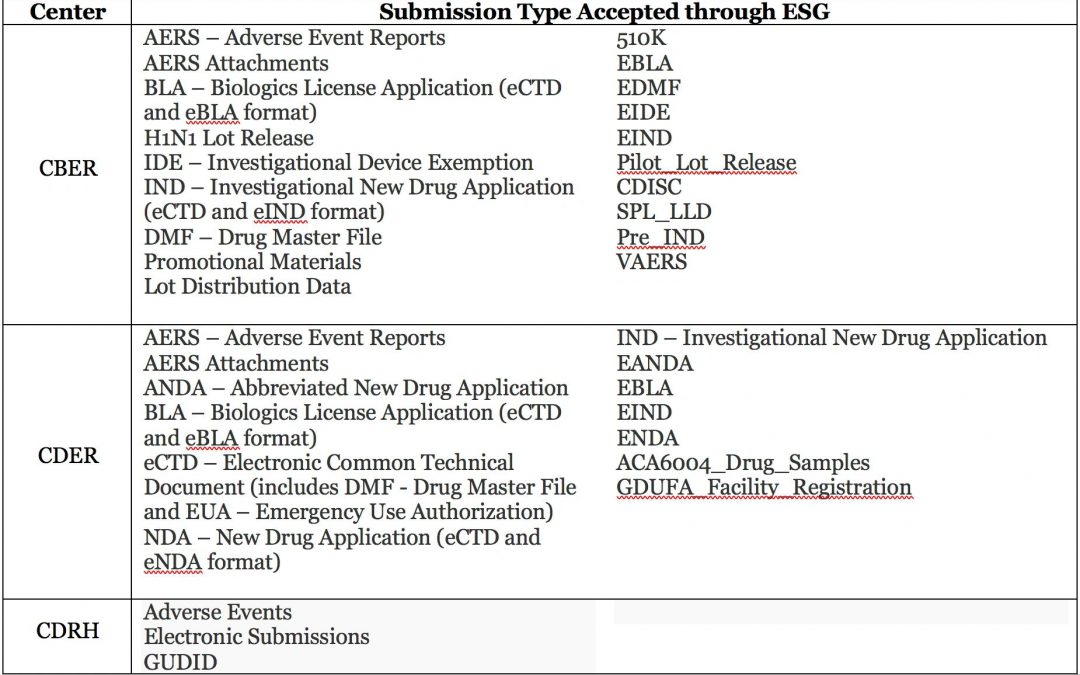
What It Is and Why We Need It Table of ESG Submission Types for Drugs and Devices The FDA’s Electronic Submissions Gateway (ESG) is a tool that facilitates the secure transmission of electronic submissions from industry partners to the agency. The Gateway is not part...
by Katrina Rogers | Sep 27, 2018 | Uncategorized
This month the FDA proposed a new rule to remove the requirements for medical device premarket submissions to be made on paper with multiple copies and replace them with a requirement for a single submission in electronic format (known as an eCopy). As a bit of...
by Katrina Rogers | Sep 25, 2018 | Uncategorized
Establishing Limits for Impurities in DrugsCDER issued an update to MAPP 5017.2 R1 [revised 01MAY2020, updated link as of 09DEC2020] in September to clarify the review instructions for drug substance and drug product impurity acceptance criteria. The change...
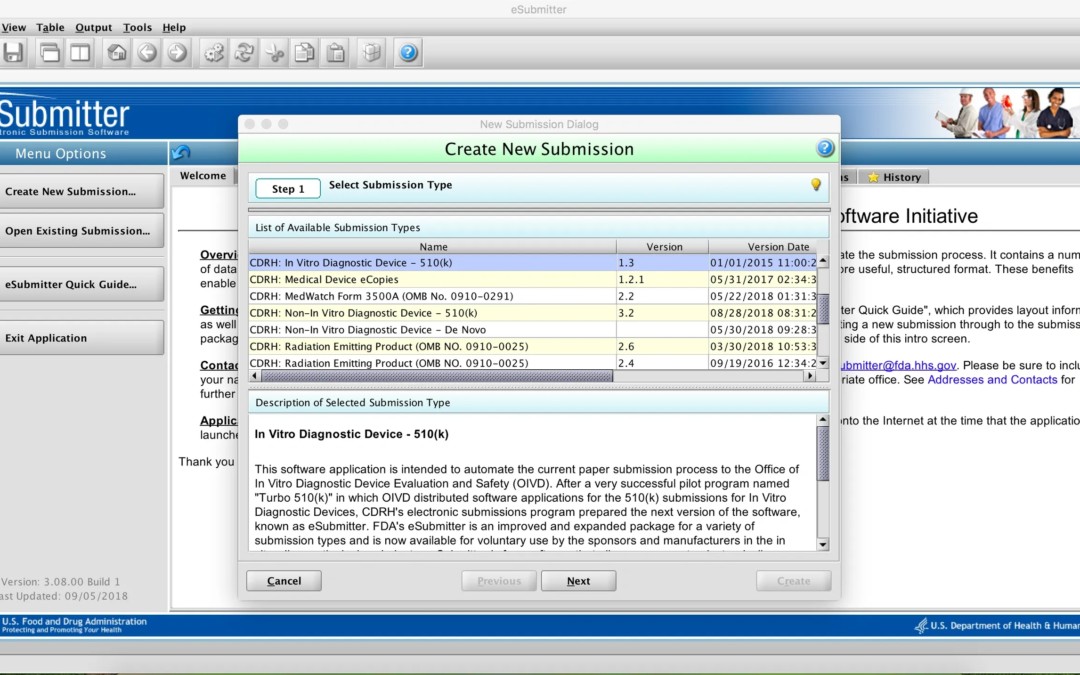
by Katrina Rogers | Sep 23, 2018 | Uncategorized
The FDA offers a free software package called eSubmitter designed to help companies prepare electronic documents for selected regulated submissions (for example, CBER Annual Reports or CDRH eMDR reporting under form 3500A). The tool creates a package of documents on...
by Katrina Rogers | Sep 20, 2018 | Uncategorized
This month the FDA posted a notice on updates to the forms for reporting adverse events related to medical products (either drugs or devices) to the agency. Manufacturers and compounders have a regulatory mandate to report serious adverse events and they use form...




Recent Comments