by Katrina Rogers | Oct 23, 2018 | Uncategorized
and what do they do?Part of the Office of Clinical Pharmacology, the Division of Applied Regulatory Science (DARS) performs applied research to “develop and evaluate novel tools, standards, and approaches to assess the safety, efficacy, quality, and performance of...
by Katrina Rogers | Oct 21, 2018 | Uncategorized
Following the release of the report “Final Report on Pharmaceutical cGMPs for the 21st Century – A Risk-Based Approach” in 2003, the Office of Generic Drugs (OGD) recognized that its process for review of ANDAs needed an update to meet the new risk-based approach and...
by Katrina Rogers | Oct 18, 2018 | Uncategorized
The Division of Industry and Consumer Education (DICE) in the Center for Devices and Radiological Health (CDRH) will be hosting a workshop for medical device manufacturers on November 6, 2018. The workshop, which has a webinar format, will focus on 2 parts of the...
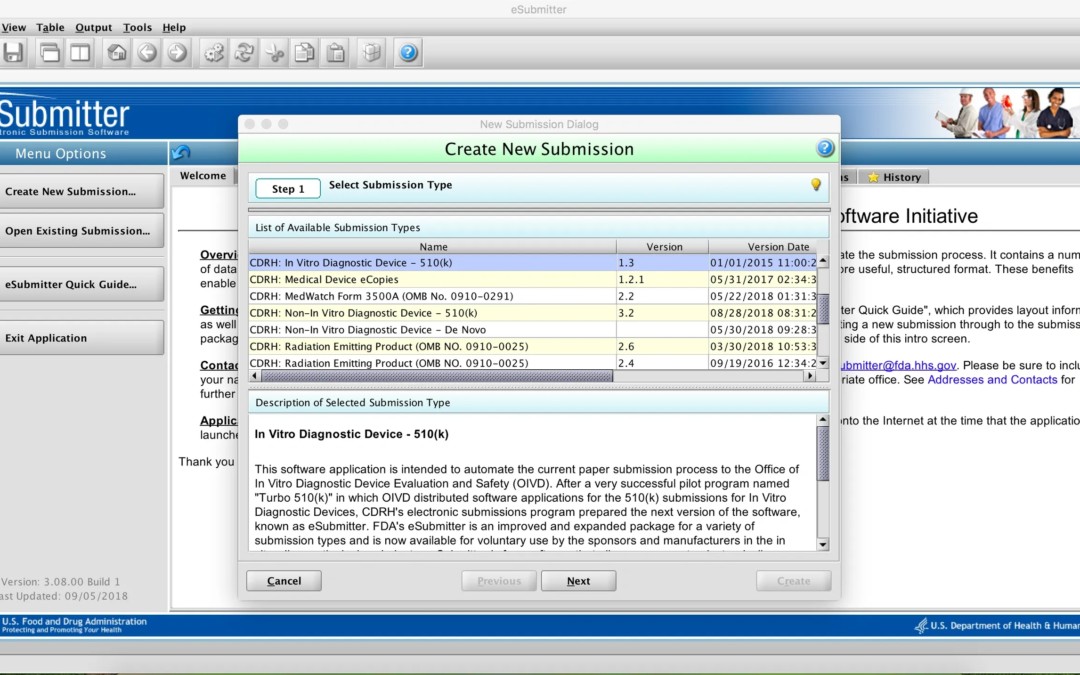
by Katrina Rogers | Oct 16, 2018 | Uncategorized
CDRH is currently running a pilot program to accept 510(k) premarket notification submissions packaged using the eSubmitter tool (see my previous post). The purpose of the pilot is to collect information to assess the value of the template in facilitating review and...
by Katrina Rogers | Oct 14, 2018 | Uncategorized
One of the current strategic priorities for CDRH is the creation of Collaborative Communities to allow interested stakeholders (both private and public, including the FDA) to work together medical device challenges. These communities may be convened by any...


Recent Comments