by Katrina Rogers | May 12, 2019 | Uncategorized
CDRH hosted a webinar on March 14th to review the current state of the Least Burdensome approach following issuance of an updated final guidance on February 5, 2019. The purpose of the approach is the remove or reduce unnecessary burden during the regulatory process...
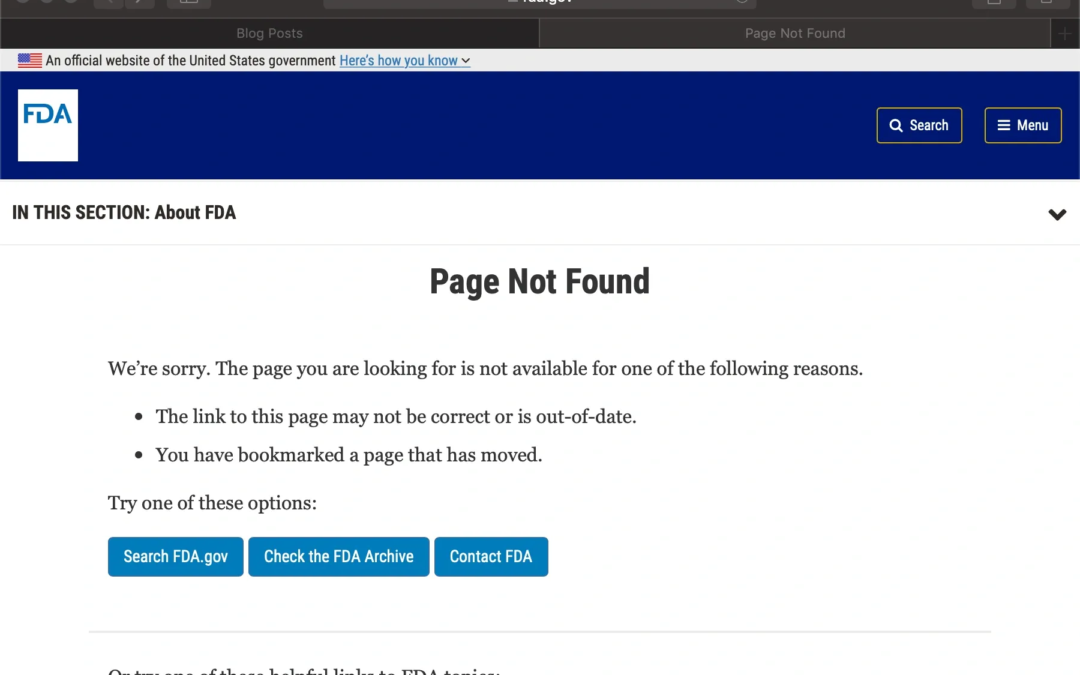
by Katrina Rogers | May 9, 2019 | Uncategorized
I’ve recently noticed some updates to the FDAs web pages that may mean some broken links in my previous posts. When you click on some links, you will see the notification shown above (see an example by clicking here). I’ve been able to find the resources by copying...
by Katrina Rogers | May 7, 2019 | Uncategorized
Readers will recall my previous post on the reorganization of CDRH to create a more adaptive structure to meet future needs. As part of that reorganization, the Office of Product Evaluation and Quality (OPEQ) was launched on May 1st, 2019 (see the announcement here)....
by Katrina Rogers | May 5, 2019 | Uncategorized
Medical Products in the NorthwestLast week’s Life Science Innovation Northwest conference offered a diverse look at the research and commercialization of medical products (both pharmaceuticals and medical devices) currently underway in the Pacific Northwest. Attendees...
by Katrina Rogers | May 2, 2019 | Uncategorized
One goal of the FDA’s Medical Device Safety Action Plan (see my previous post on the plan) is to support an improved national medical device evaluation and surveillance system. The Medical Device Epidemiology Network (MDEpiNet) is a public-private partnership...



Recent Comments