by Katrina Rogers | Jun 4, 2019 | Uncategorized
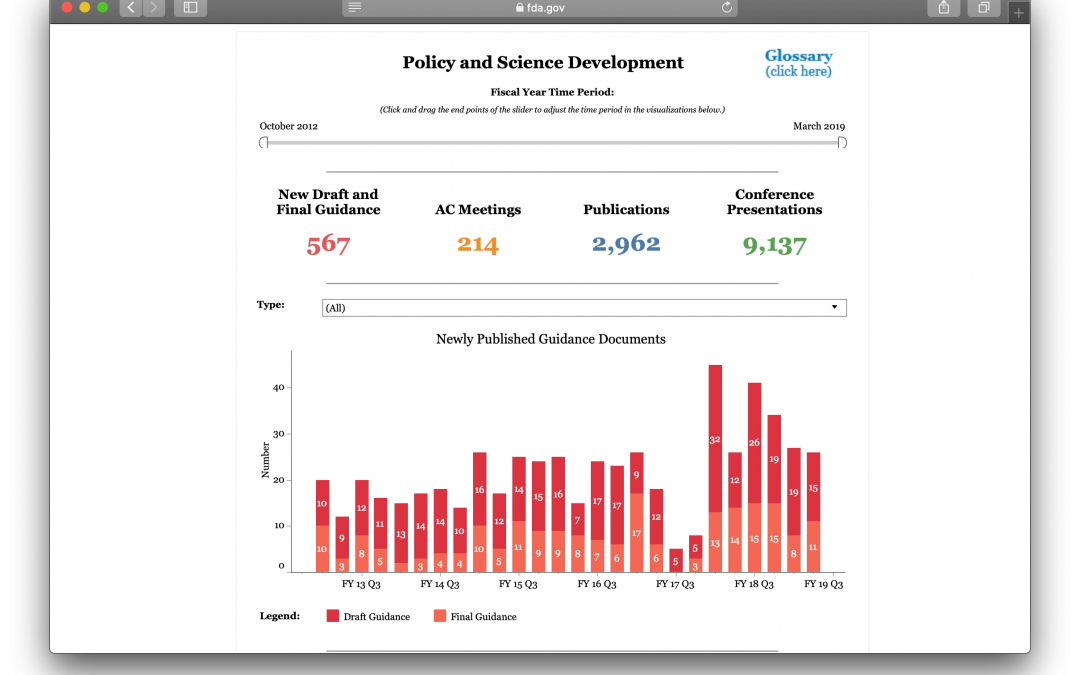
In late May CDER launched a new web page for their portion of FDA-TRACK, the performance management system used by the FDA to monitor the agencies’ programs. The new web page provides business intelligence and analytics for the division using 7 dashboards:Policy and...
by Katrina Rogers | Jun 2, 2019 | Uncategorized
It was a thought-provoking day last Thursday at the Life Science Washington East/West Life Science Summit. After hearing Amy Bernard’s call for collaboration with the Allen Institute for Brain Science, we participated in a discussion of neuroscience innovation at...
by Katrina Rogers | May 30, 2019 | Uncategorized
Readers in an around Spokane today should consider joining us to learn more about neuroscience research and development in Eastern Washington at the Life Science Washington East/West Life Science Summit. The conference started with a networking event last night and...
by Katrina Rogers | May 28, 2019 | Uncategorized
Last September the International Council for Harmonization (ICH) released its draft guideline “Nonclinical Safety Testing in Support of Development of Paediatric Medicines – S11” to provide direction on safety studies required to support the development of...
by Katrina Rogers | May 26, 2019 | Uncategorized
Practitioners of quality rely on data to determine root causes and devise effective solutions and process improvements. There are many resources available on the subject, but few are free, and rarely do we find a free resource that is as clear, usable, and...



Recent Comments