
by Katrina Rogers | Oct 29, 2019 | Uncategorized
The FDA has established 3 programs for qualification of Drug Development Tools (DDT): Animal Models, Clinical Outcome Assessments, and Biomarkers A qualified DDT is publicly available to be used in any drug development program for its qualified context of use, and...
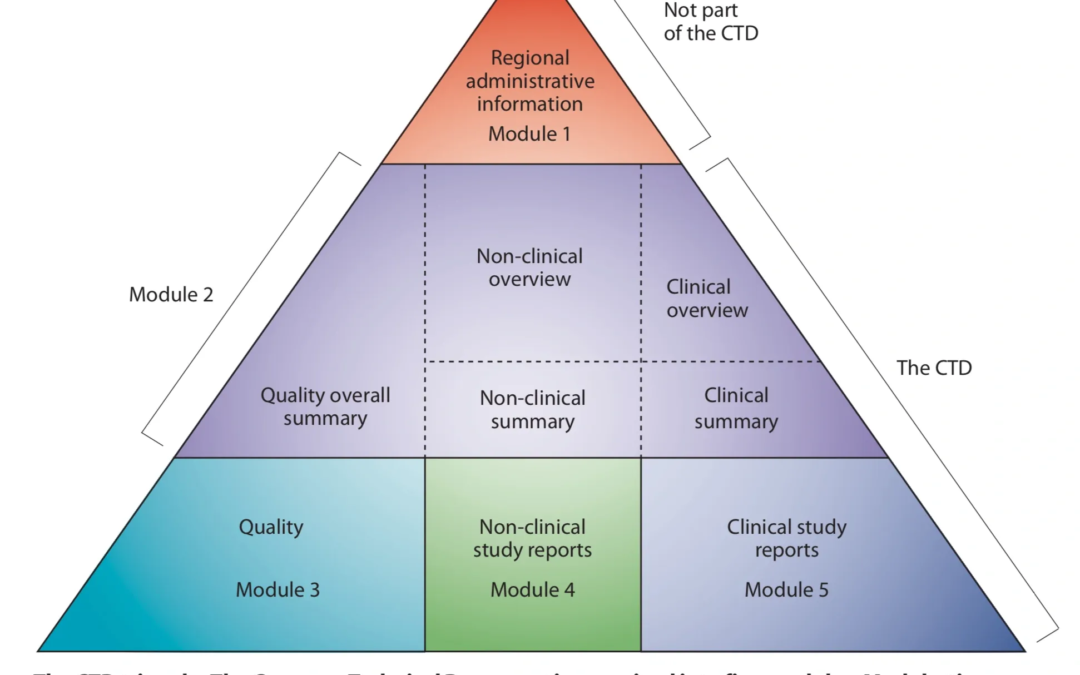
by Katrina Rogers | Oct 24, 2019 | Uncategorized
The Common Technical Document Provides a Structure for Submission (Note: This post is part of a multi-part series on the CTD this fall) Before it became electronic, the common technical document (or CTD) was proposed by an ICH steering committee as a means to...
by Katrina Rogers | Oct 22, 2019 | Uncategorized
On November 5th, CDRH will host an Industry Basics Workshop on 513(g) request for information regarding the classification of a medical device. The session will be immediately followed by another on exemptions for custom devices to address unique patient or physician...
by Katrina Rogers | Oct 17, 2019 | Uncategorized
Stay Current on Guidance When Planning Your Submissions (Note: This post is part of a multi-part series on the CTD this fall) In my last post in the series we reviewed what we could find on the history of a main eCTD guidance. There are a fair number of eCTD related...
by Katrina Rogers | Oct 15, 2019 | Uncategorized
Q&A on Evaluating Internal Standard Response In September, the CDER issued a level 2 guidance containing a series of questions and answers on the evaluation of internal standard responses during bioanalysis of clinical trial samples. Normally used in...





Recent Comments