by Katrina Rogers | Dec 3, 2019 | Uncategorized
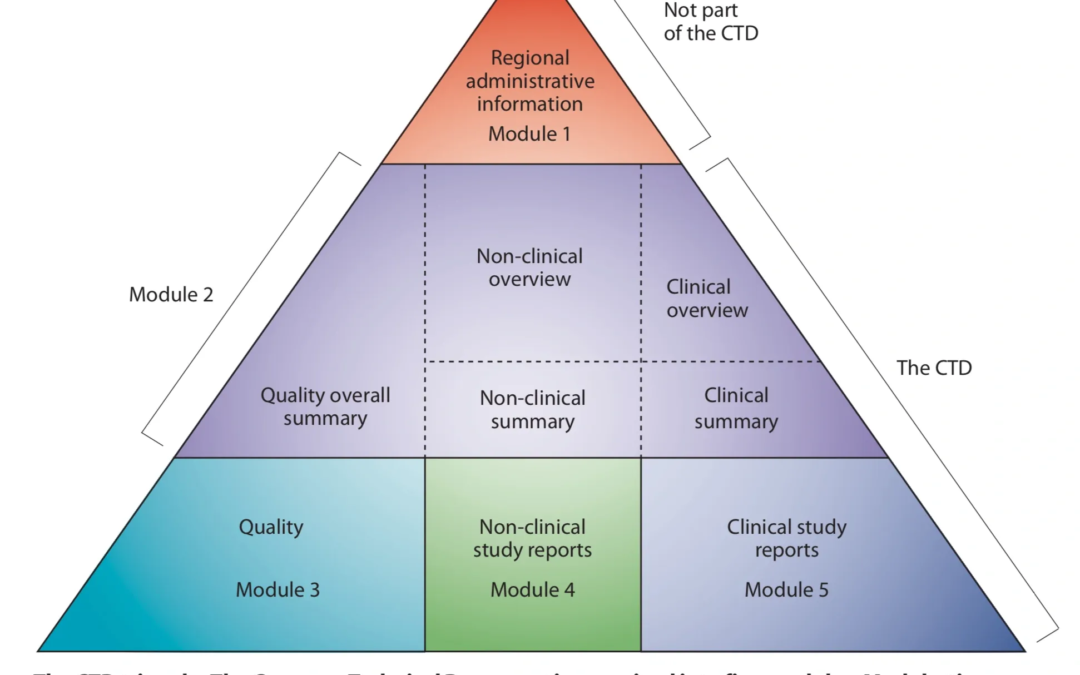
CTD Organization Makes Finding Information Simpler (Note: This post is part of a multi-part series on the CTD this fall) Part of what makes the CTD so useful is its internal structure. We can look at the Quality Overall Summary (QOS) as an example (see the M4Q...
by Katrina Rogers | Nov 28, 2019 | Uncategorized
What’s in Module 5 of the CTD? (Note: This post is part of a multi-part series on the CTD this fall) Module 5 contains the clinical study reports about the new drug. The overview and summaries for these studies are included in module 2 sections 5 and 6. Module 5...
by Katrina Rogers | Nov 26, 2019 | Uncategorized
What’s in Module 4 of the CTD? (Note: This post is part of a multi-part series on the CTD this fall) Module 4 contains the nonclinical study reports about the new drug. The overview and summaries for these studies are included in module 2 sections 4and 6,...
by Katrina Rogers | Nov 21, 2019 | Uncategorized
What’s in Module 3 of the CTD? (Note: This post is part of a multi-part series on the CTD this fall) Module 3 of the CTD contains the Quality data, which is the detailed information on the chemistry, manufacturing process, and control strategy for both the drug...

by Katrina Rogers | Nov 19, 2019 | Uncategorized
Check out new guidance and webinar for details As I’ve previously mentioned, section 3060 of the 21st Century Cures Act specifically exempts certain types of software functions from FDA regulatory oversight as medical devices. CDRH has issued the final level 1...




Recent Comments