by Katrina Rogers | Apr 28, 2020 | COVID
Chapter 13 – NIH Foundation is Coordinating a Global Research Effort This month, NIH launched a public-private partnership to develop an international strategy for the research on the COVID-19 pandemic. Part of the “part of the whole-of-government,...
by Katrina Rogers | Apr 23, 2020 | Product Development
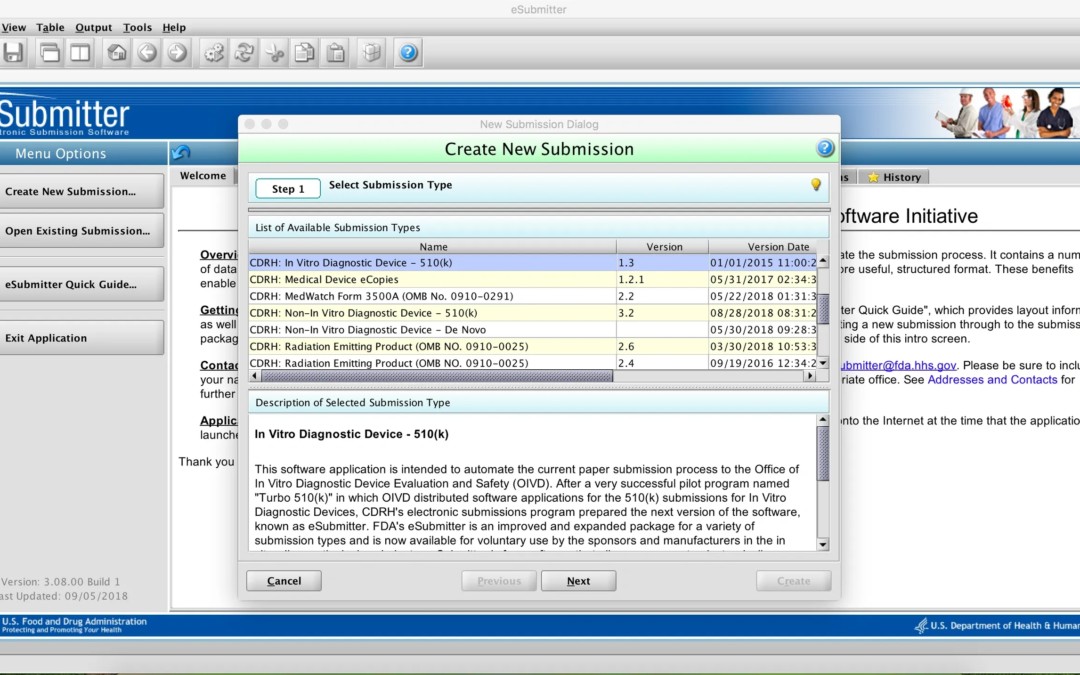
eSubmitter Tool In December, the FDA issued a final rule revising all the regulations for medical device regulatory submissions to replace paper with electronic copies. An update to the eCopy Guidance was issued with the rule, along with updates to guidance on...

by Katrina Rogers | Apr 21, 2020 | COVID
Chapter 12 – Quarantine Reading List – Leadership It’s clear we will need innovation and leadership to overcome the health and economic challenges posed by the pandemic to our businesses and community. While you are staying home to stay healthy, make...
by Katrina Rogers | Apr 16, 2020 | Medicine
Recent evidence points to an increased attention on women’s health issues at the FDA. During the FDA’s 2019 Science Forum held in September, Dr. Beverly-Lyn Cook of the National Center for Toxicological Research discussed sex and gender differences in health and...
by Katrina Rogers | Apr 14, 2020 | COVID
Chapter 11 – CDER Creates a Single Site for Its COVID-19 Response Activities So much new information is being released daily by the FDA in response to the COVID-19 outbreak that it can be hard to keep track of the latest developments or current guidance. Some...






Recent Comments